Structured Product Label Management Market Set to Exceed $210 Billion by 2035 with Veeva Systems, IQVIA, and Oracle
Structured Product Label Management Market Size, Share & Forecast to 2036
ROCKVILLE, MD, UNITED STATES, March 26, 2026 /EINPresswire.com/ -- The global structured product label management (SPLM) market is rapidly emerging as a mission-critical layer in regulated industries, where accuracy, compliance, and speed-to-market define competitive advantage. According to Fact.MR-aligned industry analysis, the market is projected to expand significantly, reaching multi-billion-dollar valuation by 2035, supported by strong double-digit growth driven by digital transformation.For Details Deep insights, Please Request A sample report for Free: https://www.factmr.com/connectus/sample?flag=S&rep_id=14637
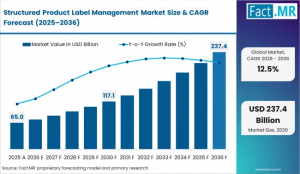
Recent estimates indicate the market is expected to grow from ~USD 2.1–6.4 billion in 2025 to as high as USD 3.4–210 billion by 2035, depending on scope definitions, with a CAGR ranging between ~6% and 12.8%+ across segments.
This signals a powerful shift—where labeling is no longer a documentation task, but a strategic digital capability embedded in product lifecycle management.
Quick Stats That Matter to Decision Makers
Market Size (2025): ~USD 2.1B – 6.4B
Forecast Value (2035): Up to USD 3.4B – 210B (scope-dependent)
CAGR: ~6% to 12.8%+
Core Industry: Pharmaceuticals & Life Sciences
Leading Segment: Software (~75% share)
Top Vendors Share: ~70% controlled by top 5 players
Why SPL Management Is Becoming Strategically Critical
In regulated industries, labeling errors are not operational issues—they are compliance risks.
Structured Product Label Management enables organizations to:
Standardize product labeling in XML-based formats (SPL)
Ensure regulatory compliance (FDA, EMA, global agencies)
Automate submission, updates, and lifecycle tracking
Maintain audit-ready, centralized documentation systems
As regulatory complexity increases globally, SPLM is becoming the backbone of compliant product commercialization.
Market Drivers: What’s Fueling High-Growth Adoption
1. Rising Regulatory Pressure
Global agencies mandate structured, machine-readable labeling formats, pushing companies toward automated SPL systems.
2. Digital Transformation in Life Sciences
Pharma and biotech firms are adopting cloud-based platforms to streamline compliance workflows and reduce manual errors.
3. Need for Faster Time-to-Market
Automated label creation and submission significantly reduce approval timelines, enabling quicker product launches.
4. Increasing Complexity of Global Labeling
Multi-language, multi-region regulatory requirements demand centralized, structured content systems.
Segment Insights That Define Market Strategy
By Component
Software (~75%) dominates due to automation, compliance tracking, and integration capabilities
Services support implementation, validation, and consulting
By Application
Pharmaceuticals & Biopharma: Core demand drivers
Medical Devices & CROs: Growing adoption for regulatory submissions
By Deployment
Cloud-based solutions are gaining traction due to scalability and real-time collaboration
Competitive Landscape: Platform Ecosystems Take the Lead
The market shows high concentration among leading regulatory tech providers, including:
Veeva Systems
IQVIA
PharmaLex
ArisGlobal
Oracle Health Sciences
Top vendors collectively control ~70% of the market, driven by end-to-end regulatory platforms and AI-enabled compliance tools.
Emerging players are focusing on:
AI-driven document structuring
XML automation and conversion
Modular compliance solutions
Regional Outlook: Growth Expanding Beyond Mature Markets
North America: Largest market due to FDA-driven compliance requirements
Europe: Strong adoption aligned with EMA standards
Asia Pacific: Fastest-growing region with expanding pharma manufacturing and regulatory alignment
Analyst Perspective
The SPLM market reflects a deeper transformation:
Labeling is evolving from static documentation to dynamic, structured, and intelligent data systems that power regulatory compliance and global product strategy.
Strategic Takeaways for Decision Makers
Invest in cloud-based SPL platforms to ensure scalability and compliance
Leverage AI-driven automation to reduce errors and accelerate submissions
Standardize global labeling processes for multi-market expansion
Partner with regulatory tech providers for integrated lifecycle management
Browse Full Report: https://www.factmr.com/report/structured-product-label-management-market
Purchase Full Report for Detailed Insights
For instant access to this report, click "Buy Now" or connect with our analyst for customization: https://www.factmr.com/checkout/14637
Have a specific Requirements and Need Assistant on Report Pricing or Limited Budget please contact us - sales@factmr.com
Related Fact.MR Reports
Structured Cabling Market: https://www.factmr.com/report/1030/structured-cabling-market
Product Launch Software Market: https://www.factmr.com/report/product-launch-software-market
Product Engineering Services Market: https://www.factmr.com/report/3756/product-engineering-services-market
Production Page Volume Market: https://www.factmr.com/report/1445/production-page-volume-market
S. N. Jha
Fact.MR
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.